
Public urged not to buy or consume unregistered pharmaceutical
Public urged not to buy or consume slimming product of unknown composition and unregistered pharmaceutical products (with photos)

Lax social media policies fueled a prescription drug boom - Protocol

FDA Advisory No.2023-0160

Making medicines trustworthy: pharmaceutical technologies of trust and regulation - Allegra Lab

Public urged not to buy or consume unregistered pharmaceutical products (with photos)

FSA issues warning on fake chocolate brands

Fighting the illicit trafficking of cultural property: a toolkit for European judiciary and law enforcement

Are dietary supplements too loosely regulated?

Department of Health - Press Release - Public urged not to take slimming product with undeclared western medicines - 091002

U.S. says Giuliani not above the law, cannot block review of phones
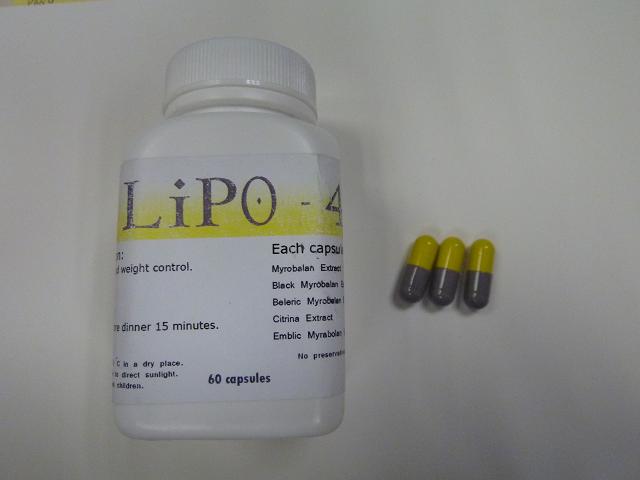
Department of Health - Press Release - Public urged not to consume slimming products with undeclared drug ingredient - 100401

Bristol Myers Squibb/Celgene Myeloma Drugs: Patient-Safety or Profit Motive? Part 1 - 340B Report

Big Pharma Plays Hide-the-Ball With Data

Six Democratic and Republican U.S. Senators Ask 340B Stakeholders for “Bipartisan Policy Solutions” - 340B Report

Federal Government Passes on Appealing Key Patient Definition Ruling - 340B Report








