![Difference between Strong and Weak Base - with Examples [in Table]](https://d1avenlh0i1xmr.cloudfront.net/0e323ff3-079c-4d5e-b21f-0f4c25082521/differences-between-strong-and-weak-bases-01.jpg)
Difference between Strong and Weak Base - with Examples [in Table]
Strong BaseWeak BaseThey get completely ionized (split up into ions) in water and produce large amounts of hydroxide ions.These only get partially ionized (split up into ions) in water and produce less amount of hydroxide ions.pH value is close to 14 but smaller than it.pH value is closer to 7 but

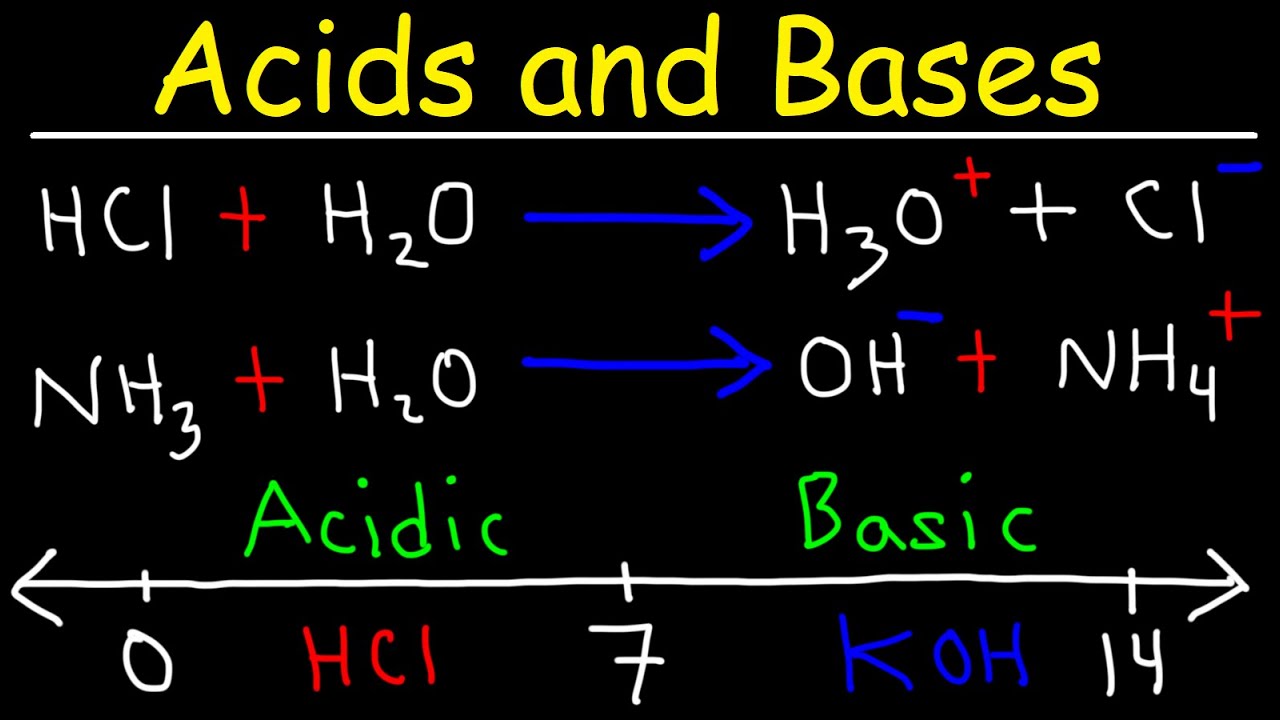
Strong Acids & Bases: Definition, Facts, Equations & Examples

Difference Between Strong and Weak Bases Definition, Properties, Reactions, Examples

Bronsted Lowry Acid and Base Theory
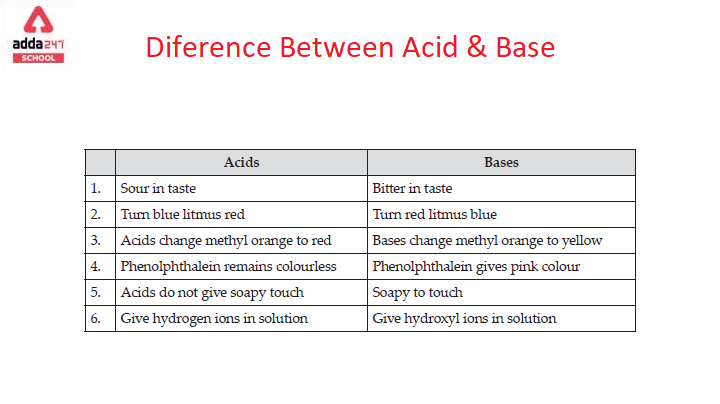
Difference Between Acid and Base for Class 7
Which indicator is used in a strong acid versus a strong base solution? - Quora

Facts About Acids and Bases

Strong vs. Weak Bases - What's the difference? How do they dissociate? The 8 Strong Bases

2.5: Weak Acids and Weak Bases - Chemistry LibreTexts
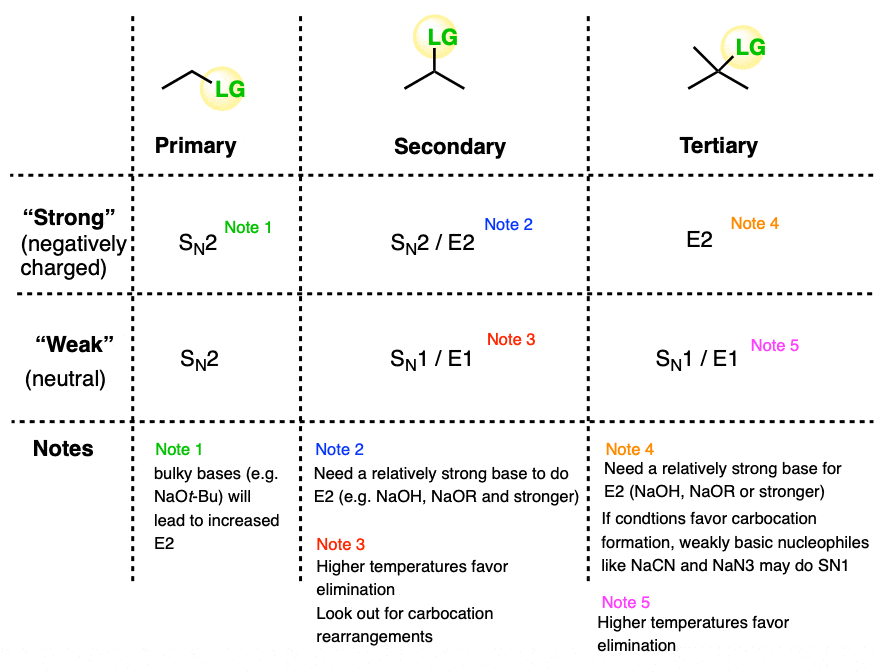
Deciding SN1/SN2/E1/E2 (2) - The Nucleophile/Base
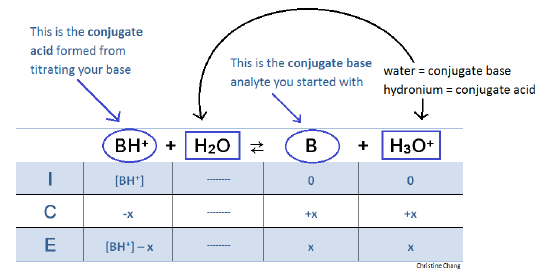
Titration of a Weak Base with a Strong Acid - Chemistry LibreTexts
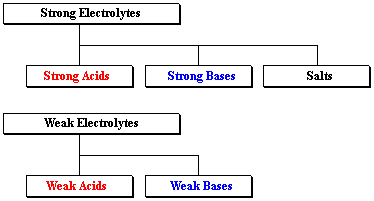
Classifying Electrolytes

physical chemistry - What is the pKa Range for weak acids and bases? - Chemistry Stack Exchange

SN1 SN2 E1 E2 - How to choose the coorect mechanism