
What Is Regulatory Dossier and What Does It Contain? - The

EU Regulatory Pathways for ATMPs: Standard, Accelerated and Adaptive Pathways to Marketing Authorisation: Molecular Therapy Methods & Clinical Development

Medical Writing, Regulatory Writing Basics

Product Dossier Registration Process - Pharma Beginners

A PRESENTATION ON DOSSIER.pptx
Costa Rica regulatory process for medical devices

Dossier Management and Regulatory Affairs.

What is Dossier in Regulatory Affairs?💊📋📃, Arun Kumar posted on the topic
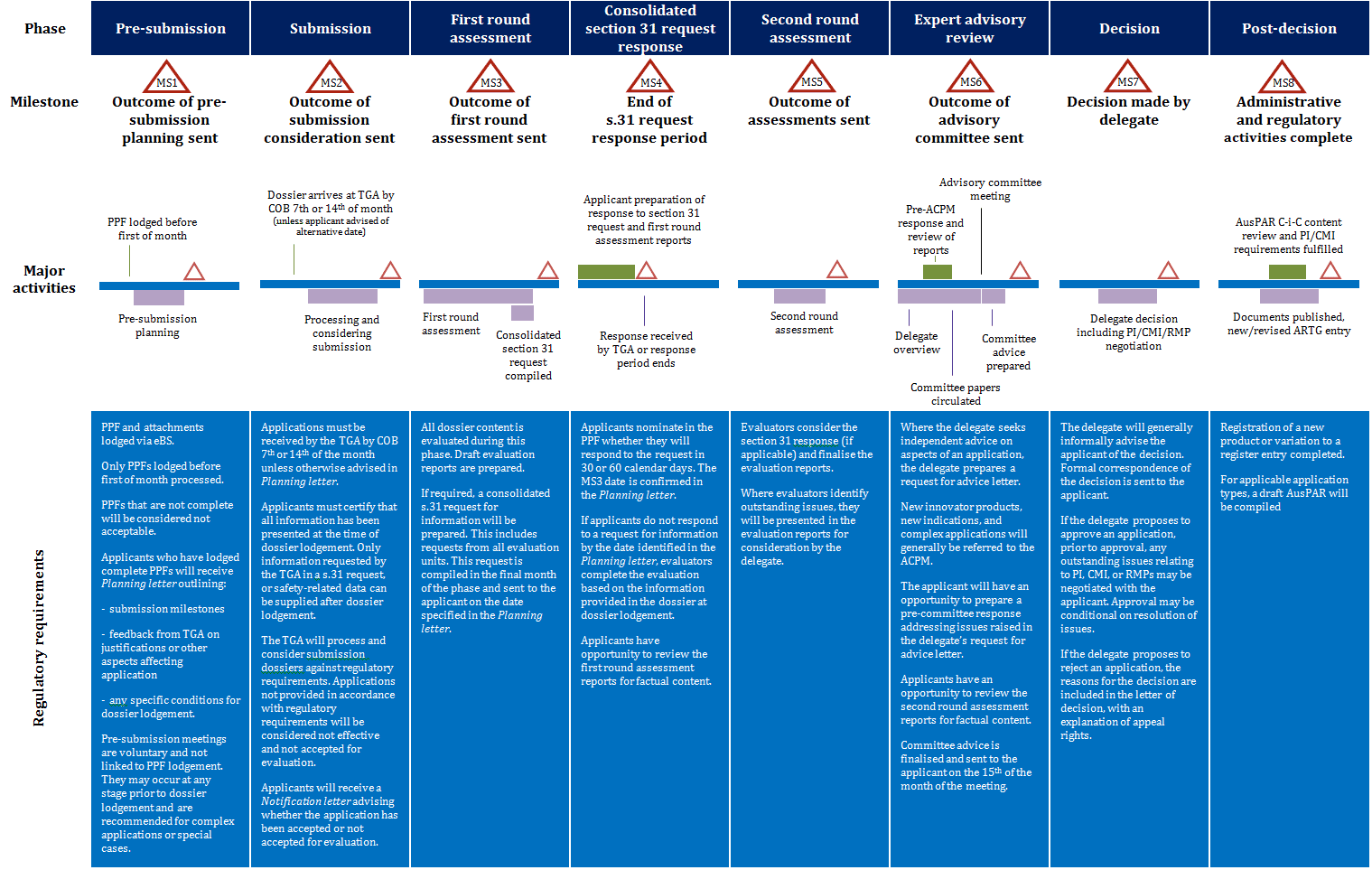
Prescription medicines registration process

Regulatory dossier preparation and submission as per CTD format

Regulatory - Famar Group

What Is Regulatory Dossier and What Does It Contain? - The Kolabtree Blog
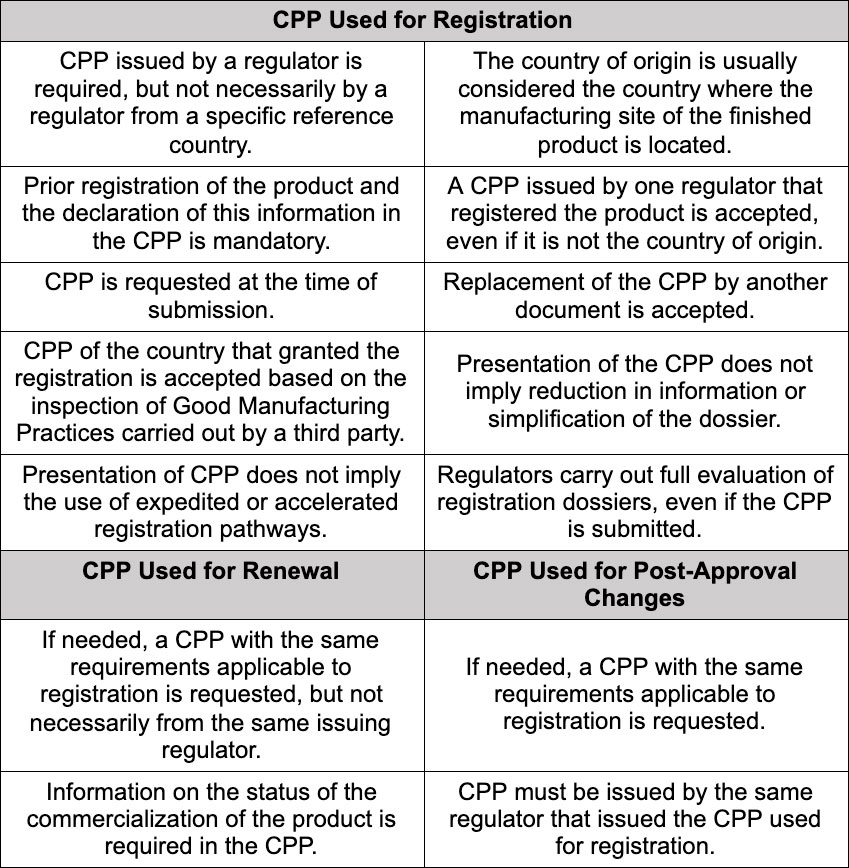
CPP Requirements for Drug Registration Processes in the Americas

Dossier Requirements for submission of new biological product.



/product/06/691714/1.jpg?0832)



