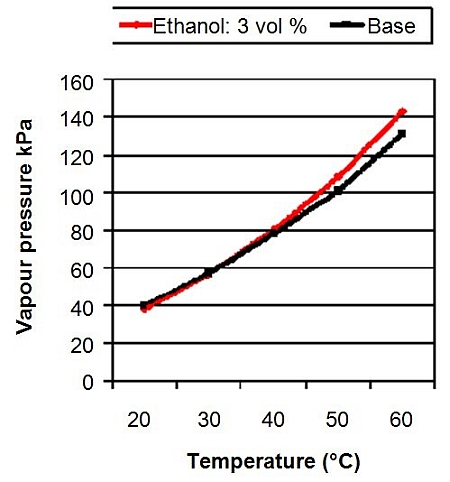
![PDF] The Addition of N-Butanol in Ethanol-Isooctane Mixture to Reduce Vapor Pressure of Oxygenated-Gasoline Blend | Semantic Scholar PDF] The Addition of N-Butanol in Ethanol-Isooctane Mixture to Reduce Vapor Pressure of Oxygenated-Gasoline Blend | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e6a7f0fc5c572d4f95f7ceb32442beee71add756/3-Table4-1.png)
PDF] The Addition of N-Butanol in Ethanol-Isooctane Mixture to Reduce Vapor Pressure of Oxygenated-Gasoline Blend | Semantic Scholar

SOLVED: 24. A solution of n-heptane and n-octane has a total pressure of 22.77 tOrL; The vapor pressures of n-heptane and n-octane are 45.8 torr and 10.9 tQrL; respectively at 25*C. (b) (

On mixing, heptane and octane form an ideal solution. At 373 K, the vapour pressure of the two liquid components (heptane and octane) are 105 kPa and 45 kPa respectively. Vapour pressure
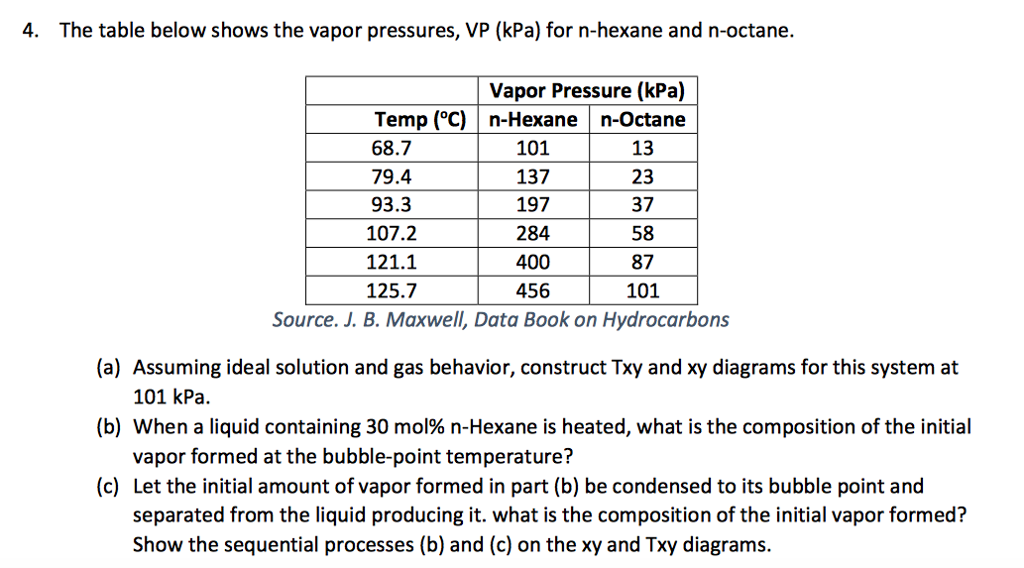
OneClass: For Problems 2 and 3, use the n-hexane, n-octane data from Problem 1. Number 1 answer is sh...
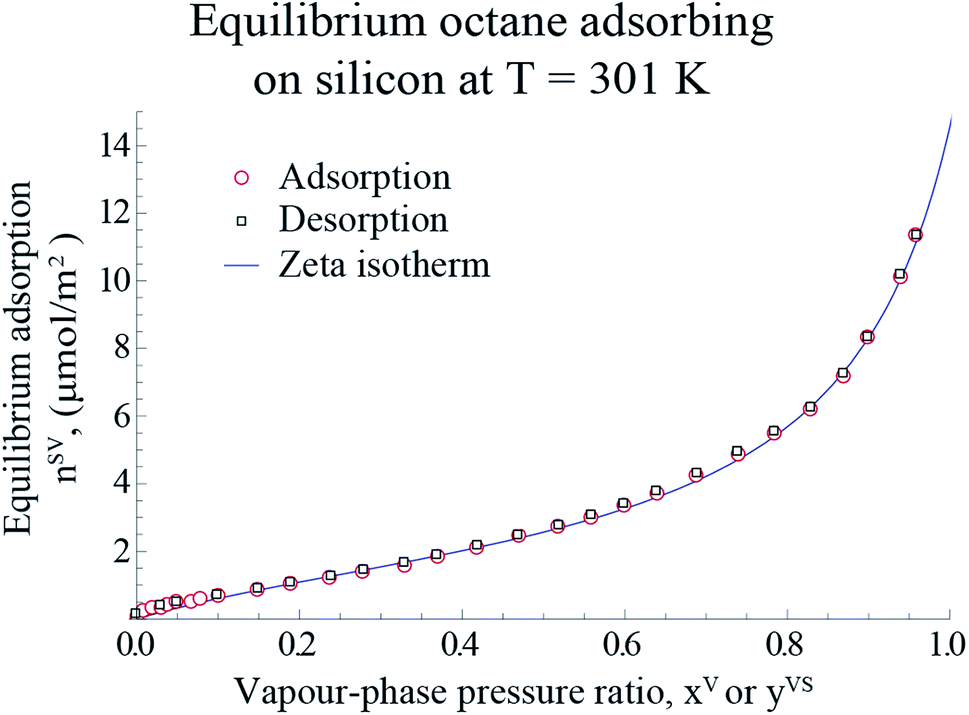
Initiation of condensation of toluene and octane vapours on a Si surface - RSC Advances (RSC Publishing) DOI:10.1039/D0RA01219J
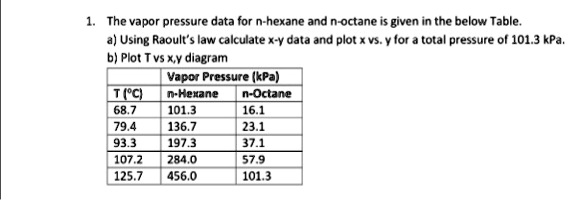
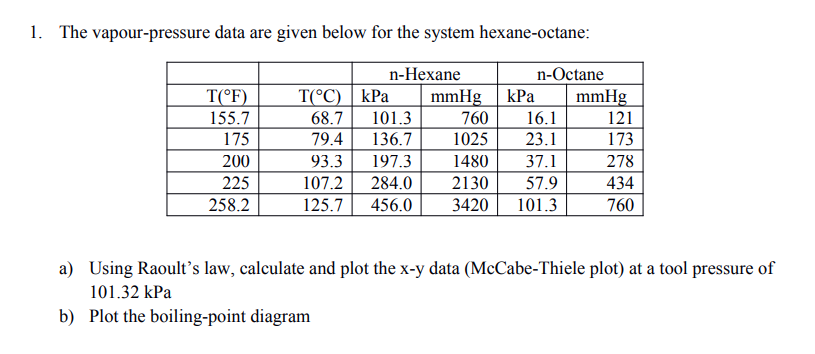
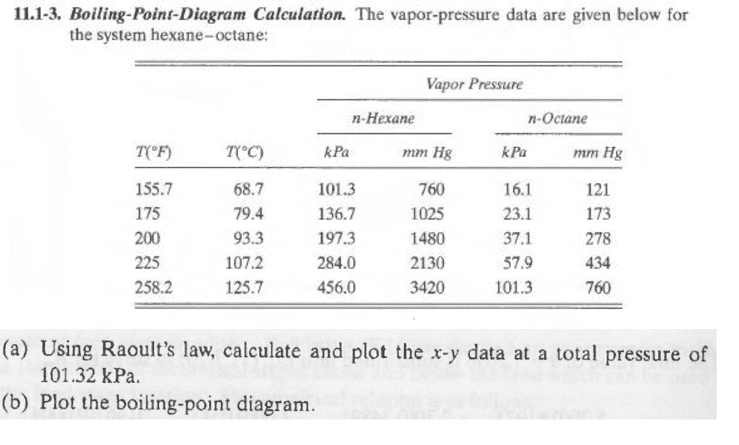
SOLVED: The vapor pressure data for hexane and n-octane is given in the below Table 4) Using Raoult'Iaw calculate X-Y data and plot * vS. lor = total pressure ol 101.3 kPa;
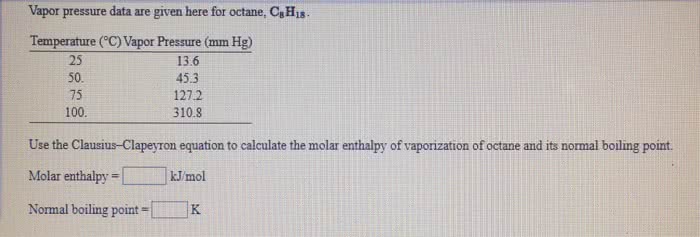
OneClass: Vapor pressure data are given here for octane, CsHis Temperature (C) Vapor Pressure (mm Hg)...

Phase equilibrium in n-octane/water separation units: vapor pressures, vapor and liquid molar fractions
2,2,4-Trimethylpentane (Iso-octane). Vapor Pressure, Critical Constants, and Saturated Vapor and Liquid Densities. | Industrial & Engineering Chemistry

6 The vapor pressure curves of pure carbon dioxide. Experimental data... | Download Scientific Diagram

Lab for Vapor Pressure of a Solution - General Chemistry | CHM 115 | Lab Reports Chemistry | Docsity
![PDF] Predicting the Enthalpy of Vaporization and Calculating the Entropy of Vaporization of 87 Octane Gasoline Using Vapor Pressure | Semantic Scholar PDF] Predicting the Enthalpy of Vaporization and Calculating the Entropy of Vaporization of 87 Octane Gasoline Using Vapor Pressure | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/85d2251bcd002fae24b5e90e74ffa2bba6bde5af/5-Table1-1.png)