Capsule Formulations Can Help Speed Up Drug Development Timelines
Capsule formulations are a useful and popular way to create a dosage form that can be developed in a shorter timescale than other dosage forms. Additionally, they are easy to manufacture and are well-known and well-accepted by regulatory bodies.
Tight development timelines and accelerated approval pathways favor simple, cost-effective capsule formulations.
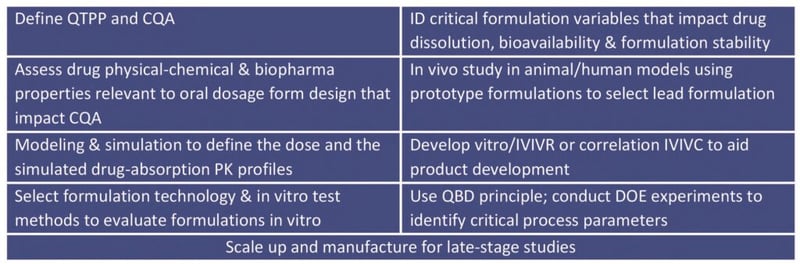
How Various Clinical Study Stages Influences Oral Formulation Approach

Making Medicines: Speeding The Path From Idea To Patient

Streamline end-to-end drug development and manufacturing - European Pharmaceutical Manufacturer
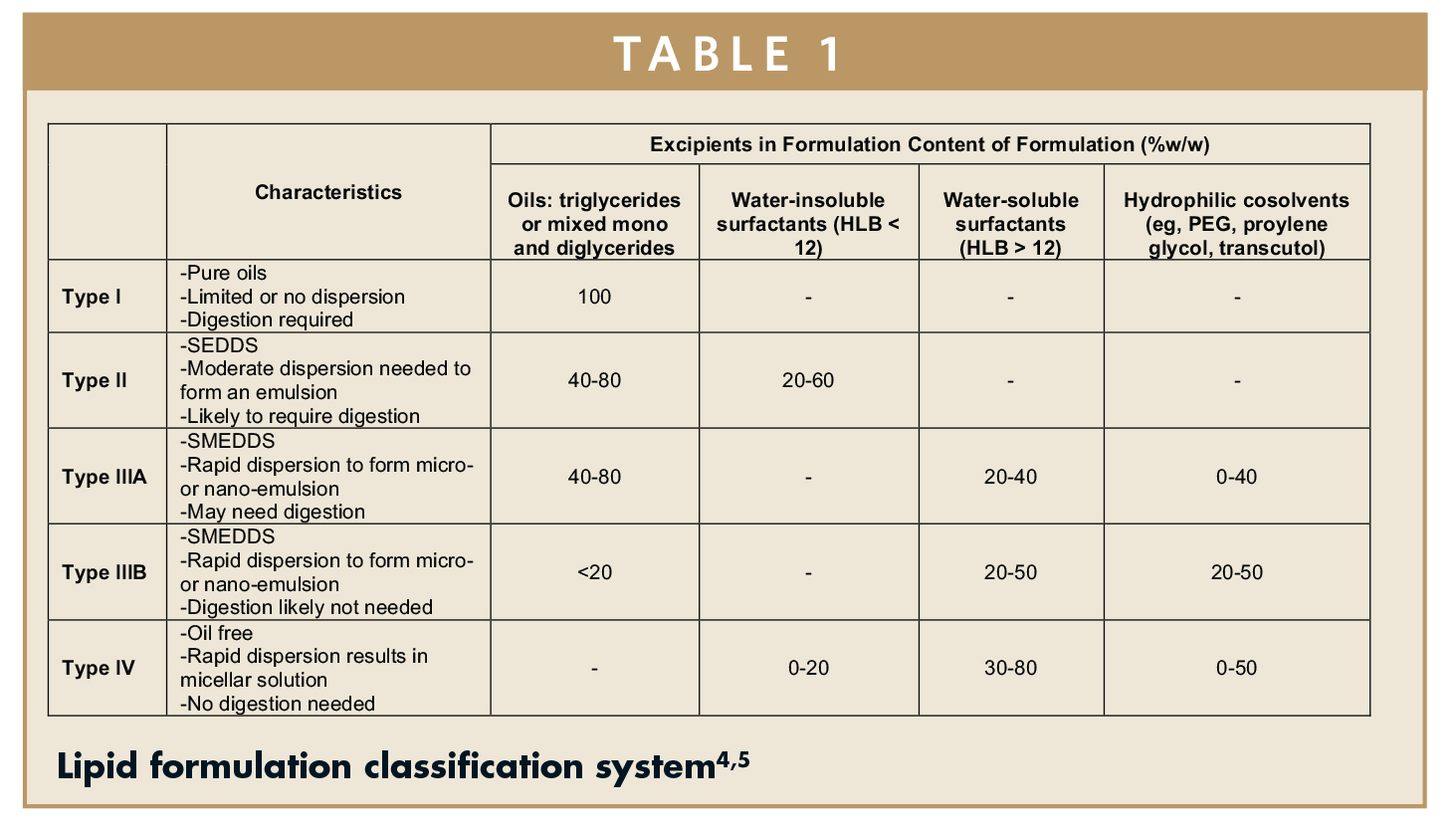
SOFTGEL FORMULATIONS - Lipid-Based Drug Delivery System to Bring Poorly Soluble Drugs to Market

Pharmaceuticals, Free Full-Text

Capsule Formulations Can Help Speed Up Drug Development Timelines
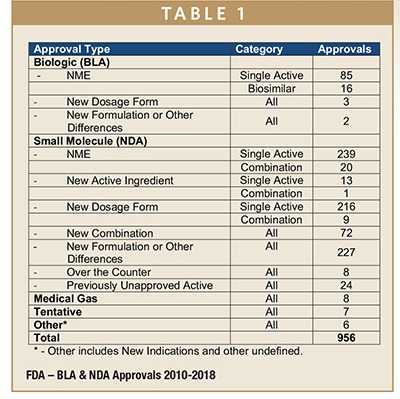
DEVELOPMENT TIMELINES - Drug Development Times, What it Takes - Part 1
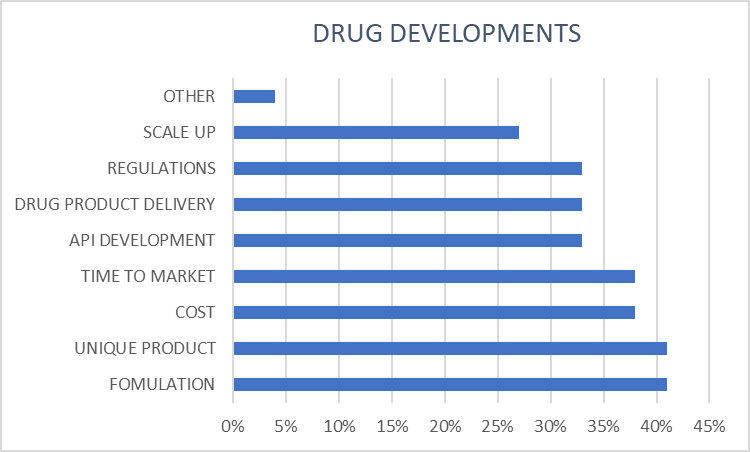
Drug Formulation Development and Clinical Trials, an Abbreviated Guide for Biotech Companies - Tayana Solutions

Application of API-in-Capsule Best Practices to Accelerate Drug Product Development

Approaches of formulation bridging in support of orally administered drug product development - ScienceDirect
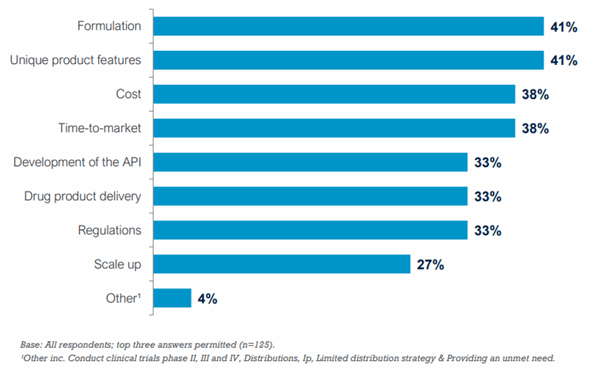
Drug Formulation Development and Clinical Trials: A Quick Guide for Biotech Companies - Sofpromed

The Lifecycle from Drug Development Through Approval Processes American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

Phase-Appropriate CMC Activities Facilitate the Transition from Early Development through Commercialization

6 Time-Saving, Tech-Driven Methods for Speeding up Drug Development - ZAGENO