
H2O Lewis Structure - Drawing Method of H2O Lewis Structure, Molecular Geometry of H2O, Polarity and Hybridisation in H2O molecule, with FAQs
A Lewis Structure is a simplified representation of the valence shell electrons in a molecule. Water is made up of two hydrogen atoms and one oxygen atom. Dihydrogen monoxide is another chemical name for this molecule.

Education Sciences, Free Full-Text

Lewis Dot Structure for H2O (Water)

H2O Lewis structure, Molecular geometry, Bond angle, Shape

H2O Lewis Structure - Drawing Method of H2O Lewis Structure, Molecular Geometry of H2O, Polarity and Hybridisation in H2O molecule, with FAQs

H2O Lewis Structure - Drawing Method of H2O Lewis Structure, Molecular Geometry of H2O, Polarity and Hybridisation in H2O molecule, with FAQs

H2O Molecular Geometry / Shape and Bond Angle (precise angle is 104.45)
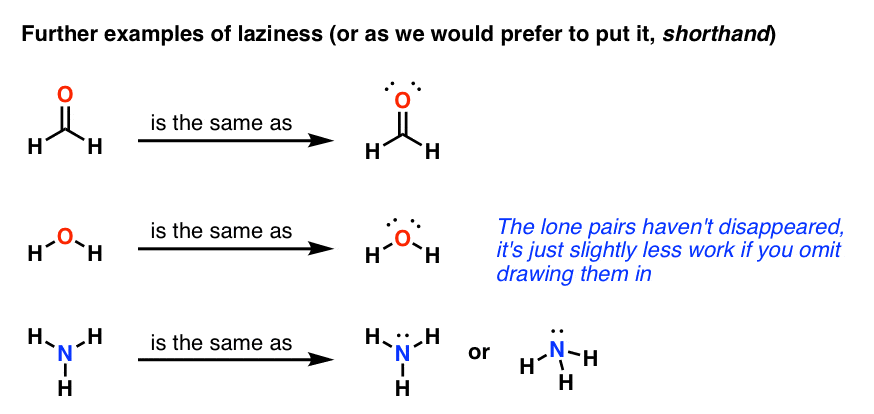
Lewis Structures – Master Organic Chemistry
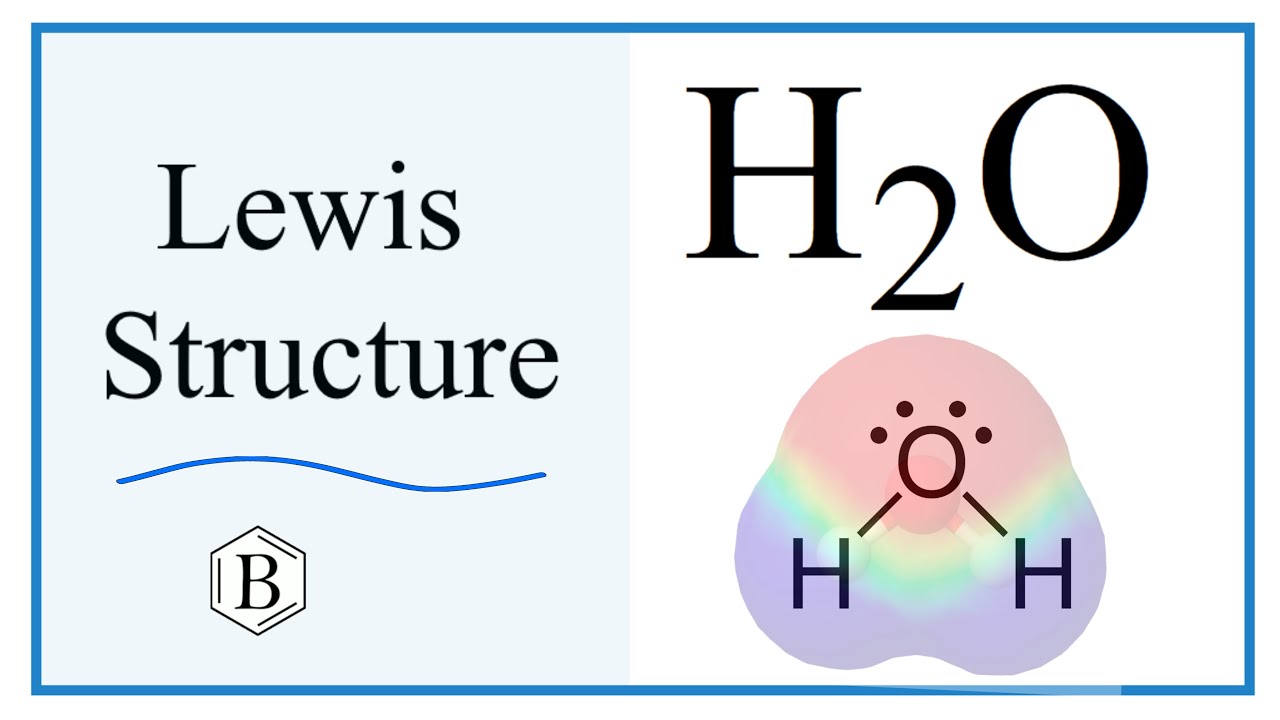
Lewis Structure for H2O
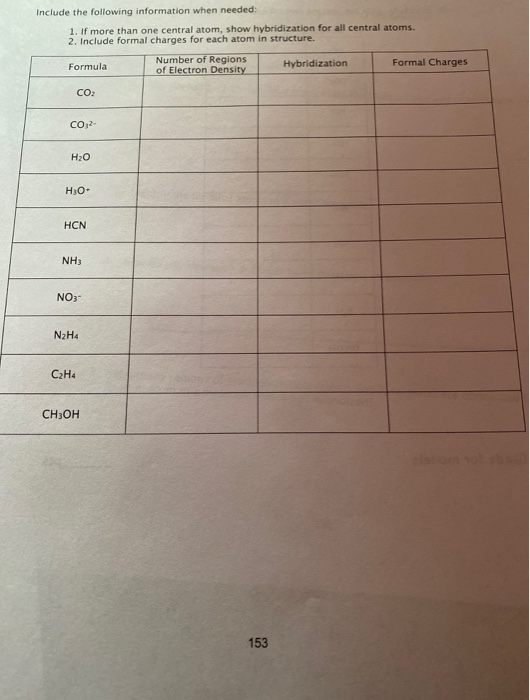
Solved Include the following information on the Lewis

Dipole & Dipole Moment, What is Molecular Polarity? - Lesson

Complete the following for H2O. a. Draw the Lewis dot structure. b. Draw a second structure with bonds drawn. c. Tell the shape using VSEPR theory. d. Tell if the molecule is

Give the Lewis dot structure of H2O.

H2O Lewis Structure: Lewis Dot Structure for H2O
Structure of H2O2









