.jpg)
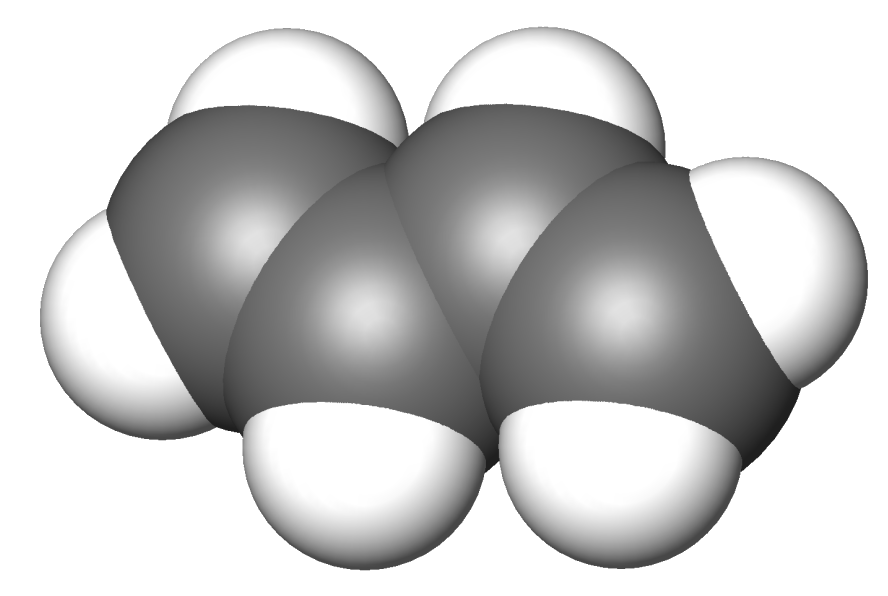
Buta-1,3-diene
Butadiene is manufactured from fractions obtained from the distillation of oil. Although by far its major use is in the manufacture of artificial rubbers
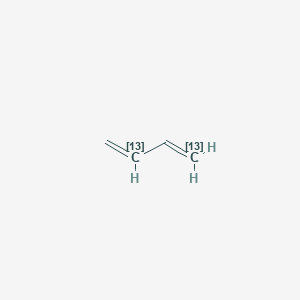
1,3-13C2)buta-1,3-diene, C4H6

Multi-Component Synthesis of a Buta-1,3-diene-Linked Covalent Organic Framework
Butadiene - Wikipedia

buta-1,3-diene - an overview

Addition of bromine to buta-1,3-diene gives:
E)-1-(2,3,6-trimethylphenyl)buta-1,3-diene (TPB, 1)

6. Draw resonance srtuctures of thefollowing:A. PhenolB. BenzaldehydeC. Buta -1,3-diene D. Acetate ion
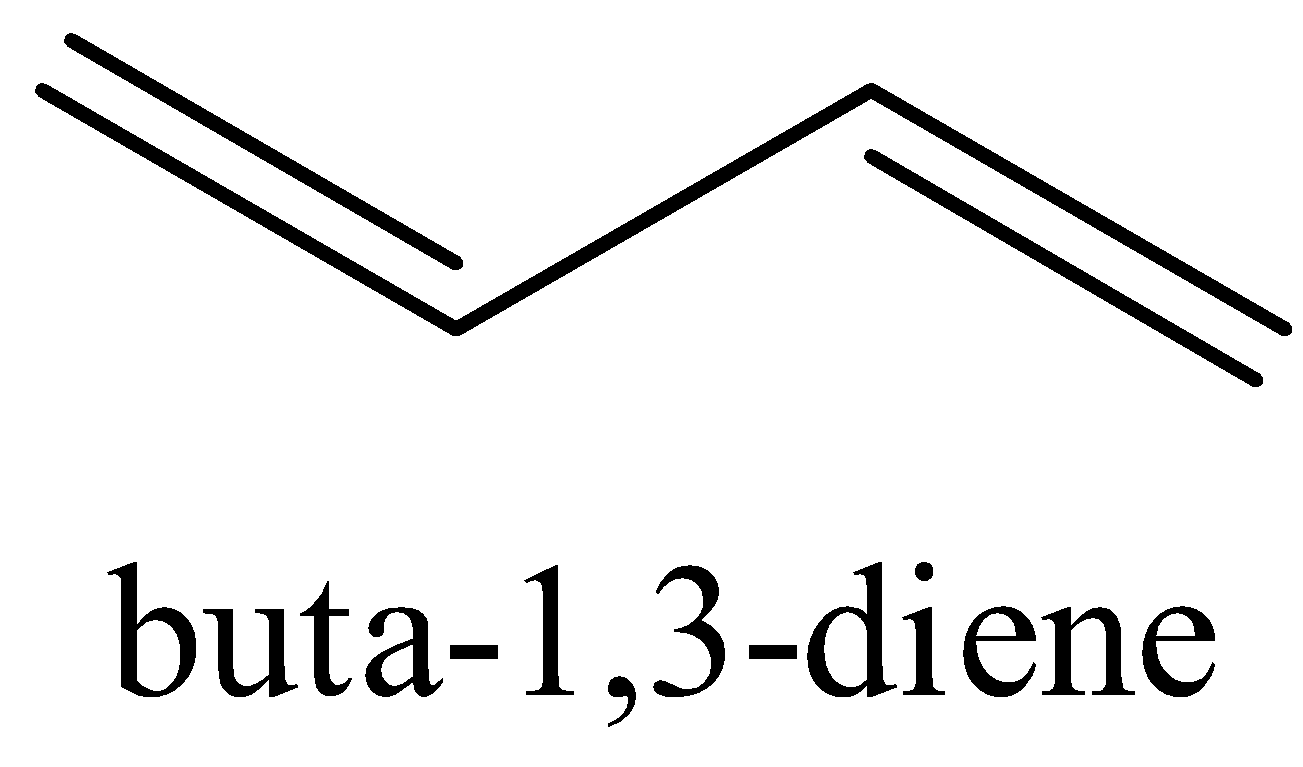
Statement I: ${{Buta - 1,3 - diene}}$ is less stable than ${{penta - 1,4 - diene}}$.Statement II: ${{Buta - 1,3 - diene}}$ has a greater number of resonating structures and the delocalized

buta-1,3-diene, in stock

1,4-Benzoquinone is a good Diels–Alder dienophile. Predict t

The compound buta 1,2 diene has
Buta-1,3-diene;but-1-en-3-yne, C8H10
.jpg)
Buta-1,3-diene